Driving enteric health through manipulation of intestinal microbial fermentation without supplementation of in-feed antimicrobial growth promoters
Introduction
Consumer- and legislation-driven pressures to remove or reduce the use of antibiotics in monogastric animal production in Asia-pacific region has been increasing, and most of the countries in the region have implemented a partial or full ban on in-feed antibiotics. Removing antibiotics in the diet changes the dynamics of the microbial ecology in the gastrointestinal tract (GIT) from a limited diversity and density to a more diverse and dense microbial community. While the industry practice with AGP has been primarily relied on controlling enteric diseases by lowering microbial density in the GIT, the target of formulation without in-feed antibiotics has to evolve to steering the dense GIT microbial community to a less harmful and more productive community. Numerous dietary strategies have been proposed using variable mechanisms; however, the common objectives of many of the proposed approaches are reducing protein fermentation while increasing fibre fermentation in the GIT.
Relationships between intestinal microbial fermentation and enteric diseases
The microbial community in the GIT is complex and it's role in fermentation and susceptibility to invasion by pathogenic species and subsequent evolution of enteric diseases in monogastric animals is not fully appreciated. However, several indisputable findings so far include;
1) Increasing protein fermentation increases necrosis of the enterocytes and colonocytes due to production of toxic gases such as hydrogen sulphide and ammonia, and hence increases the susceptibility to enteric diseases,
2) Increasing protein fermentation and production of resultant alkaline nitrogenous compounds increases intestinal pH which is favours development of pathogenic microbiota,
3) Increasing fibre fermentation increases short-chain fatty acid (SCFA) production which not only supplies 10-25% of the animal's energy requirement but also regulates mucosal and systemic antibody capacity through the production of secretory immunoglobulin A in the lamina propria and immunoglobulins A and G in the spleen,
4) Increasing fibre fermentation and production of SCFA decreases hindgut pH which is preferred by fibre fermenting microbiota,
5) The commensal microbiota in the GIT prefers fermentation of carbohydrates, and starvation of commensal microbiota through the limited supply of fermentable fibre can increase protein putrefaction in the GIT,
6) Most of the commensal microbiota in the GIT cannot efficiently ferment highly polymerised fibres, and this is the part of the reason why dietary fibre in the commercial diet is not efficiently fermented,
7) Limited fibre fermentation (i.e., butyric acid production) and hence excessive use of circulatory glucose for ATP generation in the enterocytes increases oxygen exudation from the enterocytes, increasing oxygen levels in the GIT. This reduces fibre fermentation as cellulolytic microbes are strictly anaerobic and increases the expression of virulence factors from pathogenic microbiota that would otherwise have remained as commensals rather than becoming pathogenic.
Based on the abovementioned findings, nutritional strategies in the absence of in-feed antibiotics for a minimal susceptibility to enteric diseases should target the reduction of undigested protein flow into the distal part of the small intestine and eventually into the large intestine, while maximising the influx of fermentable fibres (fibres with a shorter degree of polymerisation) into the large intestine. While we should target increasing fermentable fibre influx into the large intestine, it is important to ensure that we do not increase digesta viscosity and fibre fermentation in the small intestine by including excessive amounts of viscous soluble fibre in the diet. Also, minimising oxygen tension in the GIT would be a prerequisite for efficient fibre fermentation.
Reducing undigested protein flow in the GIT
Reducing undigested protein flow in the GIT will reduce protein fermentation and production of protein fermentation by-products (e.g. hydrogen sulphide, amines, ammonia, indoles and phenols) . Hence, the use of highly digestible protein sources such as milk protein and plasma, reducing the protein level of the diet with crystalline amino acid supplementation, and fine grinding of protein meals are recommended. One other important factor hindering protein digestion is the activity of pepsin in the stomach, which can be reduced by the presence of phytate and high gastric pH. Presence of phytate and its lower esters reduces the activity of pepsin through hydrogen bonding in the aqueous phase with positively charged proteins (which included pepsinogen), while high gastric pH which can arise due to excessive use of the ingredients with high buffering capacity can also inhibit activation of pepsinogen to its active form pepsin. Therefore, to limit undigested protein flow to the lower GIT, a high dose of phytase, limited use of the ingredients with high buffering capacity and organic acids in the diets are recommended.
Increasing fibre fermentation efficiency in the GIT
The microbial community in the GIT is highly competitive; accordingly, increasing fibre fermentation would relatively suppress protein putrefaction. It is well established that increasing digesta viscosity or excess inclusion of dietary fibre increases endogenous amino acid secretions, especially threonine. When fermentable fibre (fibre with a low degree of polymerisation) is limiting, many species of bacteria will switch to fermentation of these endogenous amino acids resulting in increased protein fermentation in the hindgut. Therefore, excess inclusion of non-fermentable fibre should be avoided. At the same time, it is necessary to supply fermentable fibres with a low degree of polymerisation to shift microbial fermentation to a more fibrolytic rather than proteolytic state. The lower fibre fermentation in the hindgut is not likely due to a lack of fibre, as most of the corn/SBM or wheat/SBM-based monogastric animal diets contain reasonable amounts of non-cellulosic polysaccharides (8-11%). It is because the dietary non-cellulosic polysaccharides in the major feed ingredients are highly polymerised and hence are difficult to ferment by the fibrolytic microbes. In this regards, exogenous NSP degrading enzymes, particularly xylanase as half of the non-cellulosic polysaccharides found in monogastric animal diets are arabinoxylan, can create fermentable fibre in the GIT. For example, a study by Apajalahti and Bedford (1998) demonstrated that supplementation of xylanase in a wheat-based broiler diet significantly increased shorter chain arabinoxylan in the terminal ileum, which would be available for fermentation in the large intestine by fibrolytic microbes (Figure 1, left-side graph). In grower-finisher pigs, repeated studies in the US and Europe have showed an improvement in liveability of 2% when xylanase was supplemented in the diet (Figure 1, right-side graph), which is likely an indirect effect of xylanase improving gut functionality by increasing fiber fermentation and SCFA production in the large intestine.
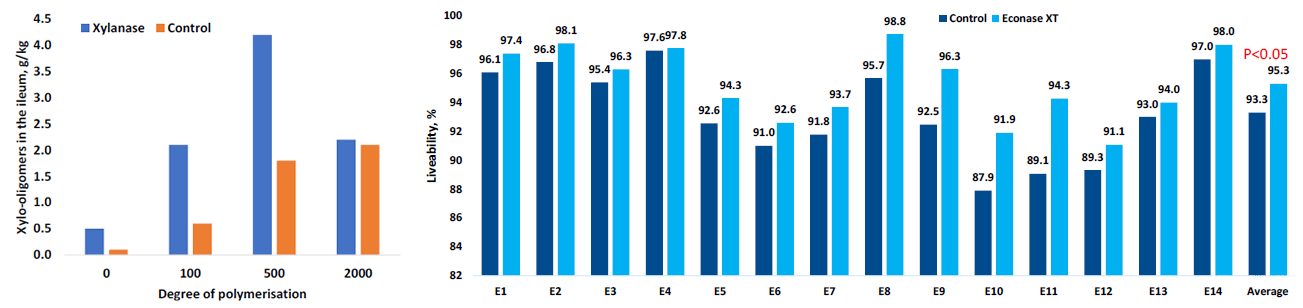
Figure 1. Xylanase increases shorter chain xylo-oligomers in the ileal digesta of broilers (Left, Apajalahti and Bedford 1998); and improves liveability (%) of grower-finisher pigs determined in the 14 US/EU commercial farms (Right, AB Vista internal study).
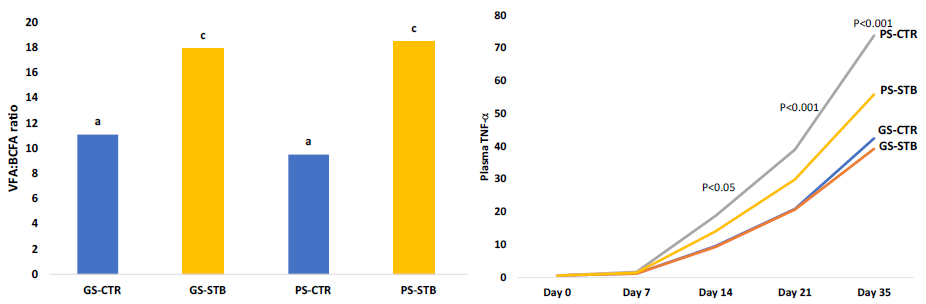
Figure 2. Supplementation of stimbiotics (STB, Signis® AB Vista) both in good (GS) and poor sanitary conditions (PS) shifted microbial fermentation pattern to more fibrolytic (higher VFA:BCFA ratio, Left); and reduced inflammatory response (TNF-a) under poor sanitary challenge (Right, Cho et al., 2019).
Reducing oxygen tension in the GIT to maximise fibre fermentation
While fibrolytic microbiota are strictly anaerobic, many of the pathogenic microbiota such as enterotoxigenic strains of E. coli and salmonella can proliferate in the presence of oxygen. Increasing oxygen tension in the small and large intestine is also associated with low fibre fermentation. Rivera-Chavez et al. (2016) examined the effect of antibiotic-mediated depletion of fibrolytic commensal microbes (Clostridia spp.) in mice. Depletion of Clostridia spp reduced butyric acid production and increased colonocyte oxygenation as colonocytes use lactic acid or glucose to generate ATP instead of butyric acid. The increased oxygen content in the intestinal lumen stimulated proliferation of aerobic pathogens such as salmonella in this study. Moreover, Sperandio (2018) elucidated in a human study that increased oxygen concentration from the microvilli capillary network into the intestinal lumen regulates oxygen-sensing transcription factors that increase the expression of virulence factors by enterotoxigenic E. coli. Therefore, one way to reduce enteric disease is to reduce oxygen tension in the intestine along with increasing fibre fermentation. Live yeast (Vistacell, saccharomyces cerevisiae) as aerobic and is known to consume oxygen and reduce oxygen tension in the intestinal tract and hence increase fibrolytic activity. Therefore, concomitant supplementation of stimbiotics and live yeast would interdependently steer microbial fermentation and contribute to maintain a healthier microbiome.
Conclusions
Without the use of in-feed antibiotics, the target for diet formulation should be a holistic approach to steer the intestinal microbiome towards more fibrolytic and less proteolytic activities. Achieving a greater fibre to protein fermentation ratio will strengthen intestinal barrier function, reduce the proliferation of pathogenic microbiota and improve performance of animals. Minimising undigested protein flow towards the lower gut, generating shorter chain fermentable dietary fibre in the small intestine, and stimulating microbial fibre fermentation in the large intestine are the strategies nutritionists should aim for to reduce susceptibility to enteric disease.
For more of the article, please click here.