Bioiberica hydrolysed porcine intestinal mucosa proteins: How to guarantee the absence of African Swine Fever Virus (ASFv)
High quality protein sources obtained by enzymatic hydrolysis of porcine intestinal mucosa could be an effective way to contain the spread of African Swine Fever virus (ASFv). With a solid supply chain and a production process certified by the Texcell-Pasteur Institute, can an ASFv free product really be guaranteed?
Currently, the industry is concerned about the biosecurity or prevention measures that manufacturers of porcine by-products can provide to ensure the non-transmission of viruses such as African Swine Fever (ASF). This disease is responsible for massive production and economic losses. It can be spread by live or dead pigs, domestic or wild, or their products, including contaminated feed or transmission through ticks, due to the high resistance of the virus in the environment.
ASFv is an enveloped virus with a double stranded DNA, approximately 170-193Kb, which codes for more than 150 proteins. Up to 34 structural proteins have been described in the ASF virus, but the P72 protein, of approximately 72,000 Daltons in weight, is the main protein of the viral capsid and it is highly immunogenic. The proteins of the ASF virus range from 10,000 to 150,000 Daltons.
Fully controlled production process
Bioiberica is a global Life Science company committed to improving human, animal and plant health and well-being. Our core business is the identification, extraction and development of animal-derived biomolecules, which are transformed into high quality products for the pharmaceutical, nutraceutical, veterinary, feed and agricultural industries.
Bioiberica is a vertically integrated company, guaranteeing its presence throughout the product life cycle from initial development and production to commercialisation. The raw material is selected and controlled at the company's eight plants in EU, the United States and Brazil. All the production facilities have implemented a common quality control system and environmentally friendly processes.
Bioiberica Animal Nutrition specialises in the development, production and commercialisation of products designed to improve intestinal and immunological health during the early stages of development of farm animals, aquaculture species and pets. The company is a leading manufacturer of hydrolysed proteins from porcine intestinal mucosa, a by-product of heparin extraction. All the raw material used to produce the hydrolysed proteins is collected from authorised slaughterhouses in the EU and certified by the sanitary authorities.
In the GMP and GMP+ production plant located in Palafolls, Spain, an ASFv-free country, the extraction process begins with the enzymatic digestion of porcine intestinal mucosa. Exclusive collection lines for porcine intestinal mucosa from healthy animals fit for human consumption are used, with exclusive transport facilities and vehicles, direct discharge in stainless steel tanks and PCR-controlled origin. The risk of cross contamination is minimised, due to systematic audits, carried out by international certification companies. All this provides a solid supply chain model and eliminates any potential presence of viral particles in the final product.
After the exclusive enzymatic hydrolysis process, the molecular weight of our hydrolysed proteins becomes less than 10,000 Da (peptides). In addition, Pasteur Institute (Texcell) validates that any potential pathogen of the porcine intestinal mucosa is inactivated with the temperature and length applied in the thermic treatment after enzymatic digestion, separation and filtration phases.
Effectively inactivating the ASF virus
Data from published literature, suggest that heat treatment is very effective in inactivating ASFv. Turner and collaborators* evaluated the heat inactivation of pig slurry with presence of ASFv indicating that inactivation of ASFv is reached at 53°C pH8 for 5 minutes, and 65°C for 1 minute. But Bioiberica goes further. Texcell, a spin-off from the Pasteur Institute, validated the heat treatment process to assess the elimination of any virus focusing in ASFv in November 2018. In Bioiberica, two thermic steps are applied:
• After enzymatic hydrolysis and before first decantation phase: 90°C±2°C for 3 hours.
• Before second decantation phase: 100°C±5°C for 1 hour, 85°C±5°C for 30 minutes.
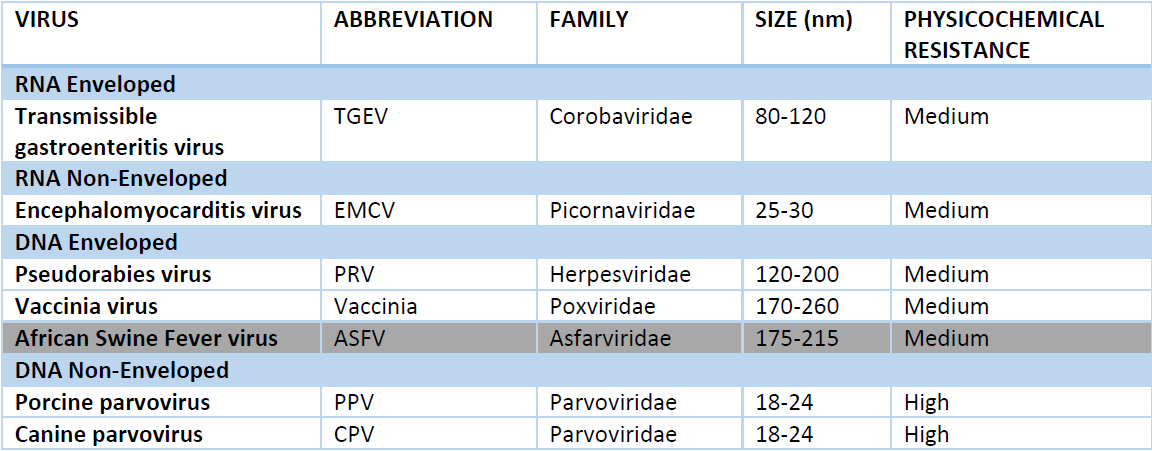
ASFv is a very particular virus. It is not commonly found in laboratories involved in processes of viral validation. However, other viruses can be studied as a model for ASFv. According to European recommendations, Poxviridae, like Vaccinia virus (dsDNA, enveloped), shares common characteristics with ASFv and thus it is an excellent virus model for a viral validation. In Texcell's technical report, both steps were studied in worst case scenario and attenuated conditions, in order to determine the kinetics of inactivation and explore its robustness.
Decantation phase 1: 90°C ±2°C for 3 hours
Vaccinia virus, Encephalomyocarditis virus (EMCV; ssRNA, non-enveloped) and Canine Parvovirus (CPV; ssDNA, non-enveloped) were used to validate this step using two types of starting material such as an intermediate of production (intestinal porcine mucosa) and a surrogate (medium alternative media). Attenuated conditions of 55°C ±1 for Vaccinia virus and 65°C±1 for CPV and EMCV, showed the total inactivation of Vaccinia virus within 3 hours of treatment and EMCV in 10 minutes. CPV was partially inactivated as it was expected, because CPV has a high physicochemical resistance compared to Vaccinia virus and EMCV with medium resistance. Results of these experiments indicated the total inactivation of all viruses tested as soon as the infected starting material reached the temperature of 87°C.
Based on Texcell's studies, total inactivation can be claimed for a medium resistant enveloped virus like ASFv, and robustness of this inactivation has been fully validated in terms of both temperature and treatment duration. Using the worst-case scenario approach, Texcell certifies that Bioiberica can extrapolate the result obtained with the most resistant virus used (PPV) to ASFv which belongs to a less resistant type of enveloped viruses. Results obtained with more sensitive viruses like PRV and TGEV using attenuated conditions, evidence a very large safety margin, in terms of both temperature and treatment duration.
Decantation phase 2: 100°C±5°C for 1 hour, 85°C±5°C for 30 minutes
Four viruses were tested: Pseudorabies virus (PRV), Transmissible Gastroenteritis virus (TGEV), Encephalomyocarditis virus (EMCV) and Porcine parvovirus (PPV), which are representative of porcine adventitious viruses covering a large range of resistance to physicochemical treatments. Scaling down of the thermal concentration step was performed in worst case conditions, with the following temperature parameters 93°C for 1h, 78°C for 30 minutes.
For the sensitive enveloped viruses PRV and TGEV, attenuated conditions were used: PRV was submitted to 45°C for 1h and then 30°C for 30 minutes, and TGEV was submitted to 60°C for 1h and then 45°C for 30 minutes. The four viruses were totally inactivated once the temperature reached 91°C.
Effect of high-quality hydrolysed proteins
This research leaves no doubt on the total inactivation of potential infectivity in the porcine intestinal mucosa in case of potential biosecurity breaches in the measures taken in order to prevent the spreading of ASF in Europe. Bioiberica guarantees that its raw material does not contain any virus as this has been eliminated in the production process. The collected filtration liquid that is finally obtained after heparin complex extraction, is concentrated to get a high-quality hydrolysed protein, which is safe, sustainable and of a high biological value. These hydrolysed proteins, known under the product name Palbio 50 and Palbio 62 are characterised by their great palatability and aroma, their high digestibility and an interesting profile of free amino acids and bioactive peptides in their composition. All of these helps to improve the digestibility of the diet and feed consumption, enhancing the productive parameters and the intestinal health of the animals.
For more of the article, please click here.
Article made possible through the contribution of Xavier Córdoba and Bioiberica